Entropy, Information, and Probability
For over sixty years, since I first read
Arthur Stanley Eddington's
The Nature of the Physical World, I have been struggling, as the "information philosopher," to
understand and to find simpler ways to
explain the concept of entropy.
Even more challenging has been to find the best way to teach the mathematical and philosophical connections between entropy and
information. A great deal of the literature complains that these concepts are too difficult to understand and may have nothing to do with one another.
Finally, there is the important concept of
probability, with its implication of
possibilities, one or more of which may become an
actuality.
Determinist philosophers (perhaps a majority) and scientists (a strong minority) say that we use probability and statistics only because our finite minds are incapable of understanding reality. Their idea of the universe is that it contains infinite information which only an infinite mind can comprehend. Our observable universe contains a very large but finite amount of information.
There is a very strong connection between entropy and probability.
Ludwig Boltzmann's formula for entropy is
S = log
W, where W stands for
Wahrscheinlichkeit, the German for probability.
As to the symbol
S, I suspect that
Rudolf Clausius, who first defined and named entropy, used
S in honor of
Sadi Carnot, whose study of heat engine efficiency showed that some fraction of available energy is always wasted or dissipated, only a part can be converted back to mechanical work.
That part of the energy that remains available to be converted to work (sometimes called "free energy") is parallel to the amount of information left in a system that is being dissipated. Entropy is a measure of the information lost to increasing chaos.
S = log
W is mathematically identical to
Claude Shannon's expression for information
I, but with a minus sign and different dimensions.
Boltzmann entropy:
S = k ∑ pi ln pi. Shannon information:
I = - ∑ pi ln pi.
Boltzmann entropy and
Shannon entropy have different dimensions (
S = joules/°K,
I = dimensionless "bits"), but they share the "mathematical isomorphism" of a logarithm of probabilities, which is the basis of both Boltzmann's and
Gibbs' statistical mechanics..
The first entropy is material, the latter mathematical - indeed purely
immaterial information.
But they have deeply important connections which information philosophy must sort out and explain.
First, both Boltzmann and Shannon expressions contain
probabilities and
statistics. Many philosophers and scientists deny any
ontological indeterminism, such as the
chance in quantum mechanics discovered by
Albert Einstein in 1916. They may accept an
epistemological uncertainty, as proposed by
Werner Heisenberg in 1927.
Today many thinkers propose
chaos and
complexity theories (both theories are completely
deterministic) as adequate explanations, while they deny ontological
chance. Ontological chance is the basis for creating any information structure. It explains the variation in species needed for Darwinian evolution. It underlies human
freedom and the
creation of new ideas.
In statistical mechanics, the summation ∑ is over all the possible distributions of gas particles in a container. If the number of distributions is
W , and the probability of all distributions is the same, the p
i are all equal to 1/
W and entropy is maximal:
S = k ∑ 1/
W ln 1/
W, so
S = k ln
W.
In the communication of information,
W is the number of possible messages. If the probability of all messages is the same, p
i are identical,
I = - ln
W. If there are
N possible messages, then
N bits of information are communicated by receiving one of them.
On the other hand, if there is only one possible message, its probability is unity, and the information content is
1 ln
0 = zero.
If there is only one possible message, no new information is communicated. This is the case in a
deterministic universe, where past events completely
cause present events. The information in a deterministic universe is a constant of nature. Religions that include an omniscient god often believe all that information is in God's mind.
Note that if there are no
alternative possibilities in messages, Shannon (following his Bell Labs colleague
Ralph Hartley) says there can be no new information. We conclude that the creation of new information structures in the universe is only possible because the universe is in fact
indeterministic and our futures are
open and
free.
Thermodynamic entropy involves matter and energy, Shannon entropy is entirely mathematical, on one level purely
immaterial information, though it cannot physically exist without a source of "negative" thermodynamic entropy, as
Erwin Schrödinger .taught in his famous essay "What Is Life?"
It is true that information is neither matter nor energy, which are conserved constants of nature (the first law of thermodynamics). But information needs matter to be embodied in an "information structure." And it needs ("free") energy to be communicated over Shannon's information channels.
Boltzmann entropy is intrinsically related to "negative entropy." Without pockets of negative entropy in the universe (and out-of-equilibrium free-energy flows), there would no "information structures" anywhere.
Pockets of negative entropy are involved in the
creation of everything interesting in the universe. It is a
cosmic creation process without a creator.
Visualizing Information
There is a mistaken idea in statistical physics that any particular distribution or arrangement of material particles has exactly the same information content as any other distribution. This is an anachronism from nineteenth-century
deterministic statistical physics.
 |
 |
 |
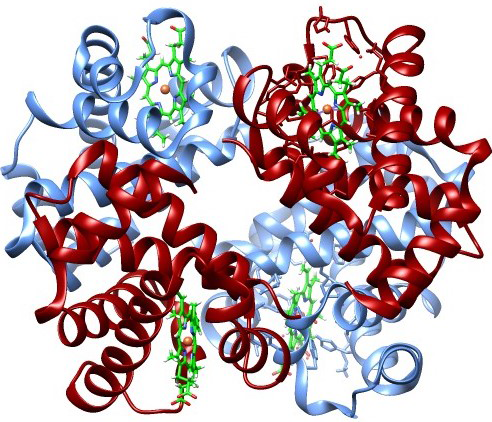
| Hemoglobin |
Diffusing |
Completely Mixed Gas |
If we measure the positions in
phase space of all the atoms in a hemoglobin protein, we get a certain number of bits of data (the x, y, z, v
x, v
y, v
z values). If the chemical bonds are all broken allowing atoms to diffuse, or the atoms are completely randomized into an equilibrium gas with maximum entropy, we get different values, but the same amount of data. Does this mean that any particular distribution has exactly the same information?
This led many statistical physicists to claim that information in a gas is the same wherever the particles are,
Macroscopic information is not lost, it just becomes
microscopic information that can be completely recovered if the motions of every particle could be reversed.
Reversibility allows all the gas particles to go back inside the bottle.
But the
information in the hemoglobin is much higher and the disorder (entropy) near zero. A human being is not just a "bag of chemicals," despite plant biologist
Anthony Cashmore. Each atom in hemoglobin is not merely in some volume limited by the uncertainty principle ℏ
3, it is in a specific quantum cooperative relationship with its neighbors that support its biological function. These precise positional relationships make a passive linear protein into a properly folded active enzyme. Breaking all these quantum chemical bonds destroys
life.
Where an information structure is present, the entropy is low and Gibbs free energy is high.
When gas particles can go anywhere in a container, the number of possible distributions is enormous and entropy is maximal. When atoms are bound to others in the hemoglobin structure, the number of possible distributions is essentially
1, and the logarithm of
1 is
0!
Even more important, the parts of every living thing are
communicating information - signaling - to other parts, near and far, as well as to other living things. Information communication allows each living thing to maintain itself in a state of
homeostasis, balancing all matter and energy entering and leaving, maintaining all vital functions. Statistical physics and chemical thermodynamics know nothing of this biological information.
Normal |
Teacher |
Scholar